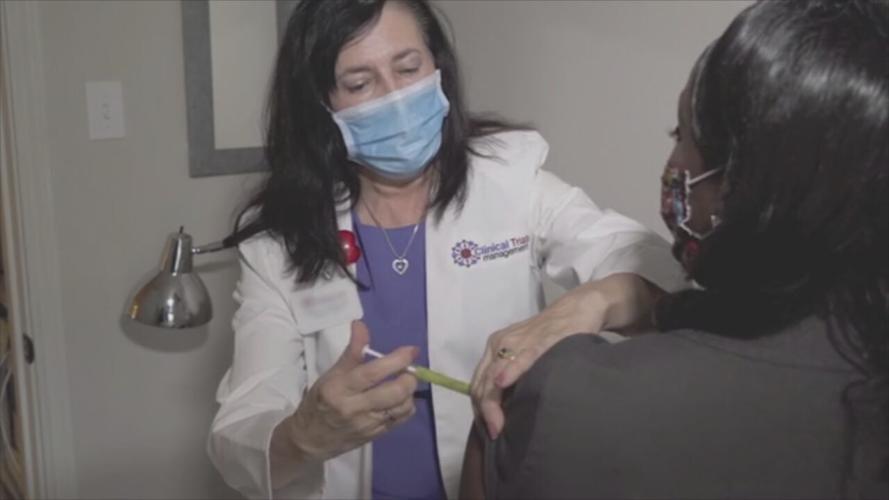
LOUISVILLE, Ky. (WDRB) -- It could be a shot in the arm for people still waiting to be vaccinated against COVID-19.
A third vaccine, developed by Johnson & Johnson and several local health care systems, could be approved for use in the United States within weeks.
Brad Strouse is a healthy 60-year-old and figured it would be a while before his number was called to get vaccinated.
"I figured it would be a long time down the road," Strouse said. "I was figuring it would be May or June before I would ever get vaccinated."
In November, Strouse volunteered to be part of Johnson & Johnson's COVID-19 vaccine clinical trials.
"After that, they sent me home, and I had no side effects," he said.
Unlike the Moderna and Pfizer shots, this vaccine candidate is a single dose and can also be stored at regular refrigeration levels.
The vaccine was tested in 45,000 people and could eventually help meet the growing demand that seems to be outpacing the current supply.
"One of the biggest problems is distribution of the vaccine," said Dr. David Dougherty, sub-investigator at Baptist Health Lexington. "And getting a single dose vaccine out, to really the world and not just this country, that can be stored in refrigerator temperatures for at least three months, is huge. The major focus should be getting as many people vaccinated as possible."
According to researchers, the Johnson & Johnson vaccine candidate is 66% effective overall in preventing moderate to severe COVID-19 infection 28 days after vaccination. But researchers say the vaccine's effectiveness increases over time across all regions studied.
"The 85% includes all those sites and if you go out to day 49 instead of 28 days status post the vaccination, there were no severe cases of COVID-19, indicating that immunity builds up over time," Dougherty said.
During a zoom press conference Friday, doctors said the efficacy rate does drop in South Africa, where many of the cases studied were due to the variant of the virus said to be more transmissible or highly contagious.
"And this new vaccine does have some efficacy against those variants," Dougherty. "It was 57% effective against preventing moderate to severe COVID-19 in South Africa."
Doctors say, if approved, the vaccine could have a significant impact on the pandemic.
"It showed complete prevention of death and hospitalization from COVID-19," Dougherty said.
"That, with a single dose, is a gamechanger," said Dr. Richard Greenberg, UK principal investigator. "It elevates the importance of this vaccine. Not just with numbers but it translates into how we as humans can survive this pandemic."
"Anything to get more vaccine at there would be a good thing," said Strouse, adding that he is glad he got the vaccine, but not just for personal reasons. "It's also good to know that it's helping others once it's approved," he said.
Johnson & Johnson plans to file an application for emergency use in the United States within a week. The company's local research partners include UK, Baptist Health Lexington and Norton Healthcare.
Copyright 2021 WDRB Media. All Rights Reserved.